An eight-step synthesis of epicolactone reveals its biosynthetic origin
07-Sep-2015
Nature Chemistry, doi:10.1038/nchem.2336
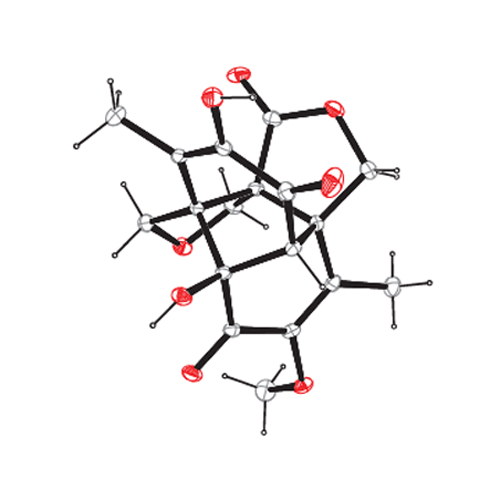
Epicolactone is a recently isolated fungal metabolite that is highly complex for its size, and yet racemic. With its array of quaternary stereocentres, high degree of functionalization and intricate polycyclic structure, it poses a considerable challenge to synthesis, a challenge that can be met by understanding its biosynthetic origin. If drawn in a certain way, epicolactone reveals a pattern that resembles purpurogallin, the archetype of ubiquitous natural colourants formed via oxidative dimerization. Based on this insight, we designed a biomimetic synthesis of epicolactone that proceeds in only eight steps from vanillyl alcohol. We have isolated a key intermediate that supports our biosynthetic hypothesis and anticipate that an isomer of epicolactone stemming from our synthetic efforts could also be found as a natural product.