Cohesin-dockerin code in cellulosomal dual binding modes and its allosteric regulation by proline isomerization
08-Feb-2021
Structure 29, 1–11, DOI: https://doi.org/10.1016/j.str.2021.01.006
Structure, online article
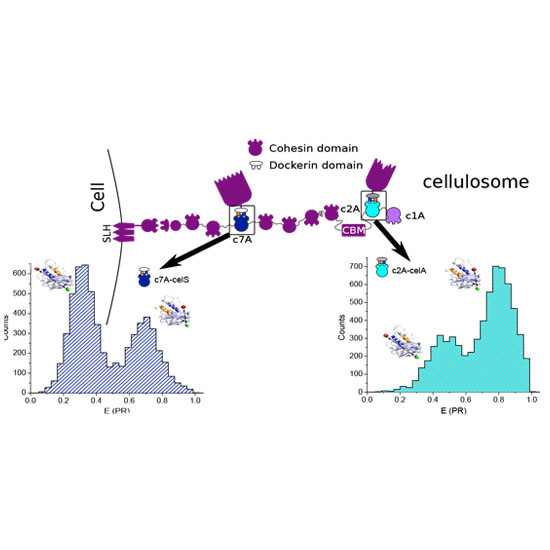
Cellulose is the most abundant organic molecule on Earth and represents a renewable and practically everlasting feedstock for the production of biofuels and chemicals. Self-assembled owing to the high-affinity cohesin-dockerin interaction, cellulosomes are huge multi-enzyme complexes with unmatched efficiency in the degradation of recalcitrant lignocellulosic substrates. The recruitment of diverse dockerin-borne enzymes into a multicohesin protein scaffold dictates the three-dimensional layout of the complex, and interestingly two alternative binding modes have been proposed. Using single-molecule fluorescence resonance energy transfer and molecular simulations on a range of cohesin-dockerin pairs, we directly detect varying distributions between these binding modes that follow a built-in cohesin-dockerin code. Surprisingly, we uncover a prolyl isomerase-modulated allosteric control mechanism, mediated by the isomerization state of a single proline residue, which regulates the distribution and kinetics of binding modes. Overall, our data provide a novel mechanistic understanding of the structural plasticity and dynamics of cellulosomes.